Overview and History of Toremifene Citrate
Toremifene (also known under its brand and trade name of Fareston) is a member of a category, family, and class of drugs that are known as SERMs (Selective Estrogen Receptor Modulators). SERMs fall under an even broader category of drugs that are known as anti-estrogens, and the cousin family of SERMs (that also fall under anti-estrogens) are aromatase inhibitors, commonly abbreviated as AIs. SERMs include compounds such as Toremifene, Nolvadex (Tamoxifen), and Clomid (Clomiphene Citrate). Aromatase inhibitors (AIs) include compounds such as Arimidex (Anastrozole), Aromasin (Exemestane), and Letrozole (Femara). Although the two fall under the category of anti-estrogens, they are both sub-categories that branch off into their own families, as SERMs and AIs differ greatly in their mechanism of action within the human body concerning how they control or block Estrogen. There has been much misunderstanding and misconception in previous decades as to what each of these do, and this should always be first clarified to the reader before describing Toremifene.
SERMs are true ‘Estrogen blockers’, while AIs are not. SERMs act to block the activity of Estrogen at their target receptor sites in different tissues (mainly breast tissue), and they do this by binding more strongly to the Estrogen receptors in these tissues than Estrogen itself, effectively taking the spot that Estrogen normally would. The SERMs will remain bound to the receptor site and remain inert (they will not activate the receptors or perform any actions) This results in the inability for Estrogen to bind to these receptors (as they are already occupied by the SERM). While SERMs block Estrogenic activity at certain receptor sites, SERMs can and do also act as estrogens themselves in other receptor sites in other tissues in the body (such as in the liver, which can be beneficial). This is known as Estrogen agonist and Estrogen antagonist activity. In any case, SERMs do not serve to lower overall total circulating blood plasma levels of Estrogen in the body – they merely block its activity in certain tissues. Aromatase inhibitors (AIs) are instead the compounds that will reduce total circulating levels of Estrogen in the body by way of binding to the enzyme responsible for the conversion (aromatization) of androgens into Estrogen. This enzyme is known as aromatase. AIs are more attractive as a substrate to the aromatase enzyme than the aromatase enzyme’s traditional substrate targets, which are androgens (such as Testosterone). The result is that the aromatase enzyme is occupied with the AI, and therefore cannot engage in chemical reactions with androgens. This results in the inability for Estrogen to be manufactured at its root source, and therefore Estrogen levels as a whole are reduced in the body.
As far as Toremifene is concerned, it is a SERM with mixed agonist and antagonist properties on the Estrogen receptor, and is classified as a triphenylethylene analogue that is very similar in structure to Nolvadex (Tamoxifen) and Clomid (Clomiphene Citrate). Toremifene (Fareston) could be considered a very close sister to Nolvadex, as they are very similar. Toremifene is also a non-steroidal compound. It is classified as a breast cancer drug in the treatment of female post-menopausal breast cancer that is determined to be Estrogen-receptor positive (or Estrogen-receptor unknown). What this means is that it is a drug that is utilized for the purpose of blocking and treating female breast cancer that is exacerbated by Estrogen via Estrogen’s activity at breast tissue receptors, which can promote cancerous growth through its tissue growth promoting activity within breast tissue. Toremifene, as previously described, binds to these receptor sites and blocks the ability for Estrogen itself to continue activating receptor sites within breast tissue, thus blocking or halting the progress of tumor growth that could be exacerbated by Estrogen.
Toremifene has gained popularity with anabolic steroid using bodybuilders and athletes for the exact same reasons that Nolvadex has: it is used to counter act and/or block some of the effects of excessive Estrogen in the body as a result of aromatization of various androgens used (such as Testosterone or Dianabol, for example). In particular, Toremifene is used to mitigate, alleviate, and/or prevent the Estrogen-related side effect of gynecomastia (the development of breast tissue on males).
Toremifene Citrate is a very new and recent compound that was approved by the FDA in 1997 as a prescription drug, and is sold most popularly under the brand and trade name Fareston on the US prescription market. GTx, Inc. was the first and current pharmaceutical company to undertake the manufacture of Toremifene under the brand name Fareston. Toremifene under the brand name of Fareston is also available in a plethora of other countries internationally as well, such as: Turkey, Thailand, Africa, Switzerland, Spain, Portugal, Russia, Sweden, Germany, Greece, Finland, Ireland, Hungary, France, South Africa, Australia, Czech Republic, Australia, Italy, and England (the United Kingdom). It is a very popular and widespread product, much like its close relative Nolvadex and can be found almost anywhere throughout the world.
The question that remains (and that is commonly asked) concerning Toremifene is whether or not it is just as effective, more effective, and/or more advantageous than its older sister Nolvadex. These questions will soon be answered here as well as in the later sections of this profile.
Chemical Characteristics of Toremifene
Toremifene (Fareston) is a non-steroidal selective Estrogen receptor modulator (SERM) that expresses both mixed agonistic as well as antagonistic properties in relation to Estrogen in different tissues and cells within the human body. Toremifene is a member of a family of compounds known as triphenylethylene compounds, which Nolvadex (Tamoxifen) as well as Clomid (Clomiphene Citrate) are both also members of, and are both in particular very closely related compounds to each other.
Properties of Toremifene
It has already been covered that Toremifene is a SERM, and serves to block Estrogen at various receptor sites in certain tissues within the body (breast tissue in particular). As a layman explanation, Toremifene pretends to be a ‘fake’ Estrogen that occupies Estrogen receptors within breast tissue. With these receptors occupied by Toremifene, real Estrogen cannot perform their jobs there. Toremifene does not reduce total blood plasma levels of Estrogen. In addition to being antagonistic to Estrogen receptors in breast tissue, it is also antagonistic to Estrogen at the hypothalamus gland (this essentially ‘tricks’ the hypothalamus into thinking there is little or no circulating Estrogen levels in the body, causing it to increase its manufacture of Testosterone so that it can utilize aromatization to restore these levels. Toremifene is also agonistic to Estrogen receptors in other tissues in the body (within the liver in particular). This means that while Toremifene will act as an anti-estrogen in breast tissue and the hypothalamus, it will act as an Estrogen within the liver. This can have beneficial impacts especially during an anabolic steroid cycle, such as improving and shifting cholesterol levels into a more favorable range.
Although Toremifene is closely related to Nolvadex, it does differ quite a bit in other aspects that have been discovered (some good and some bad). In comparison with Nolvadex, the first issue to observe is the growing concern many have towards the carcinogenic effects on liver tissue that Nolvadex exhibits. It should be understood that these effects have been observed with extremely long term use and that it should not be a problem with the very short term use that bodybuilders and athletes are engaged in, but it should be comforting for some to know that Toremifene less liver toxic/carcinogenic effects than Nolvadex[1] [2] [3], but individuals should still be aware that because Toremifene is still an analogue of Nolvadex, it does still exhibit many of the related toxic effects on the liver.
There is also clinical evidence that Toremifene may express an advantage over other SERMs such as Nolvadex through an ability to not only exhibit inhibition of breast tissue growth, but also apoptosis (or programmed cell death) of breast tissue and tumor cells in breast cancer patients[4]. This is very promising evidence and information, but it should be understood that often times breast cancer patients will react very differently from healthy individuals to different drug treatments. But this should provide some evidence that Toremifene could very well destroy gynecomastia tissue in addition to halting and preventing its growth. Toremifene (Fareston) has also demonstrated the capability to reduce Prolactin levels in the body at 300mg per day after 8 weeks[5]. This is very pleasing information, as Toremifene could possibly be utilized as an anti-Prolactin drug if needed. It is possible, however, that this might only occur in breast cancer patients.
There are some negative aspects and properties to Toremifene, however. Firstly and perhaps most importantly is the negative effect that Tore exhibits on SHBG (Sex Hormone Binding Globulin) levels in the body, where clinical research has demonstrated that while Toremifene can and does stimulate natural endogenous production of Testosterone (which is a good thing for post-cycle therapy – PCT), it in fact raises SHBG levels in the body[6]. What this means for the bodybuilder, athlete, and anabolic steroid user is that raises in SHBG will mean less free Testosterone will be present in the body. SHBG is a protein that binds to sex hormones such as Testosterone and renders it inactive while it is bound to it, in effect ‘handcuffing’ Testosterone (or other androgens and sex hormones that are susceptible to SHBG). The result is the hormone it binds to (in this case, Testosterone) ends up merely floating around in circulation unable to do its job (free vs. bound Testosterone). Elevations in SHBG are therefore not a very good thing for post-cycle recovery or for performance enhancement during cycle, which may detract from Toremifene’s potential as a recovery compound during PCT (Post Cycle Therapy). In comparison, Nolvadex has not been proven to have this effect on SHBG during the over five decades of its use and study (compared to Toremifene’s less than one and a half decade of study and use).
Furthermore, in the department of restoring HPTA (Hypothalamic Pituitary Testicular Axis) function during PCT, Toremifene has demonstrated itself to be less efficient than Nolvadex for this purpose[7]. In this study, 20mg per day of Tamoxifen (Nolvadex) over a period of 8 weeks increased LH (Luteinizing Hormone) levels upwards of 70%, resulting in an increase in Testosterone levels of 71%. In comparison, Toremifene administered over the same amount of time at 60mg per day resulted in only a 25% increase in LH levels and a 42% increase in Testosterone levels. It should be important to note, however, that 60mg per day of Toremifene is regarded as a lower dose for this purpose, but this should still clearly highlight the fact that Nolvadex remains the most potent and effective SERM among all three SERMs for this purpose, requiring the lowest dose out of all of them in order to elicit a positive effect on HPTA function. More information on suitable doses will be discussed in the Toremifene Dosage section of this profile.
Toremifene Side Effects:
Toremifene is a compound that is well tolerated by most individuals, especially by males. The most severe Toremifene side effects tend to be reported by females, especially female breast cancer patients, for whom this drug was originally intended and designated for. The reason as to why Toremifene side effects might be considered harsher or worse for females is due to the fact that Toremifene’s Estrogen agonist/antagonist properties affects women in a very different manner than males due to the simple difference in the endocrine system’s dynamics between men and women. Due simply to the fact that females possess higher Estrogen levels than males (and their bodies are more physiologically dependent on it by nature), the ramifications of Toremifene and its side effects are farther reaching and more impacting on females than in males. This is the reason why Toremifene side effects for women include things such as: abdominal or stomach pain, anxiety, change in vaginal discharge, confusion, dizziness or light headedness, hot flashes, vaginal bleeding, extreme fatigue, and other female-centric side effects. The majority of these Toremifene side effects are not experienced by males due to the hormonal differences between genders.
Clinical trials on female breast cancer patients listed the following Toremifene side effects (in order of occurrence and frequency from most experienced to least experienced): dizziness, hot flashes, sweating, elevated liver enzymes, nausea, vaginal discharge, edema, vomiting, and vaginal bleeding. Far rarer Toremifene side effects found in clinical trials using female breast cancer patients as test subjects include: stiff muscles, jaundice, breathing difficulty (shortness of breath), constipation, itching, anorexia, loss of muscle strength, depression, tremor, and visual disturbances. Note that once again, these Toremifene side effects are almost exclusively associated with females, and almost never occur in men.
For anabolic steroid using bodybuilders and athletes (who tend to be almost all males), very few negative or adverse side effects are reported. This is what makes Toremifene highly valued and sought after among anabolic steroid users, as many users have reported almost no incidence of side effects. Some users might report changes in mood (as a result of its Estrogen agonist/antagonistic activity in the body), and a positive impact in libido (likely due to its positive impact on Testosterone production). Some users have also reported a loss of libido with Toremifene, which can possibly be attributed to its SHBG increasing properties, causing less active free Testosterone to circulate in the body. Mood alteration has been anecdotally reported by many users to last only through the first several days of use before returning to normal.
There tends to be a significant lack of clinical data in regards to Toremifene side effects in males and anabolic steroid users in particular. This is because Toremifene is only about 16 years old, and the majority of its time in use has only been in a clinical setting for female breast cancer therapy. Therefore, the only current data concerning Toremifene side effects in male anabolic steroid users is in the form of anecdotal data and different experiences of users.
Toremifene Administration and Dosages:
For the treatment of breast cancer, Toremifene is used in dosages of 60mg per day. Both women and men can be prescribed Fareston (Toremifene) to help prevent the proliferation of cancer cells in breast tissues. Toremifene is usually administered for 1 year, but this can extend to 2-3 years if remission is not seen, or the decline of cancerous ductal and glandular cancerous tissues.
Steroid users and bodybuilders can use Toremifene to prevent gynecomastia due to it blocking the oestrogen receptor (ER) in breast tissue. When testosterone based anabolic steroids are administered, circulating estrogen levels increase leading to the onset of estorgenic related side effects. Toremifene is advised at 30-60mg per day, however, many opt for Nolvadex (Tamoxifen Citrate) as its a cheaper and effective alternative.
The steroid user should first look to control high estrogen levels with the usage of an aromatase inhibitor (AI), not block estrogen. Blocking estrogen is seen as a last resort of AI treatment does not suffice. When multiple androgens are used such as Testosterone Enantahte, Dianabol and Deca-Durabolin, an AI and SERM like Toremifene may be required.
Toremifene is an effective post cycle therapy (PCT) agent, but not as powerful as Tamoxifen (Nolvadex) in comparable studies. However, Toremifene is seen as a second generation SERM and a safer alternative to Nolvadex, often leading to less adverse side effects. A dosage of 120mg for the first week of Toremifene is suggested, followed by 60mg per day for 5 weeks as a post cycle treatment of low testosterone symptoms.
Buy Toremifene Online, Legality and Availability:
Toremifene Citrate (Fareston) is a prescription only drug in the Unites States, UK, Canada and Australia. That means its illegal to posses, traffic, use or manufacture. However, the drug is available by research chemical companies for “research only” and “not for human consumption” in the U.S. This means RC’s often carry Toremifene in liquid form. In liquid suspension, Toremifene is available in 30-60mg/ml and administered orally.
Many steroid sources carry SERMs, but Toremifene is rarely carried. Other SERMs such as, Clomid and Nolvadex are often in steroid suppliers inventories. Fareston is carried by few in tablet form due to it being relatively new and not favourable due to its higher price compared to other 1st generation SERMs.
A 30 tablet box of 60mg per tab Fareston will cost around $50-100. Compared to a 30 tablet box of Nolvadex or Clomid selling for $40. As a gynecomastia preventative measure or treatment, this makes it three times the price of other effective SERMs.
Using anabolic steroids can often mean serious and permanent side effects. Then obtaining ancillary medications like Fareston can be expensive and problematic. Therefore, we at Steroidal.com suggest using our sponsors to obtain safe and high quality anabolics without the legal ramifications.
Toremifene Pictures:

Farestos 20mg Pharmacom Labs

Fareston 60mg 30 Tabs GTx
Fareston 60mg Pharma
Toremifene Chemical Information:
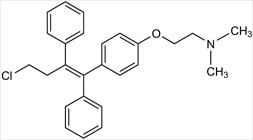 Toremifene Citrate (AKA Fareston, Acapodene)
Toremifene Citrate (AKA Fareston, Acapodene)
Chemical Name: 2-{4-[(1Z)-4-chloro-1,2-diphenyl-but-1-en-1-yl]phenoxy}-N,N-dimethylethanamine
Molecular Weight: 405.959 g/mol
Formula: C26H28ClNO
Original Manufacturer: GTx, Inc.
Half Life: 5 days
Detection Time: Unknown
Anabolic Rating: N/A
Androgenic Rating: N/A
Toremifene References:
[1] Evaluation of the genotoxicity of tamoxifen in the liver and kidney of F344 gpt delta transgenic rat in 3-week and 13-week repeated dose studies. Kawamura Y, Hayashi H, Kurata Y, Hiratsuka K, Masumura K, Nohmi T. Toxicology. 2013 Jul 30. pii: S0300-483X(13)00193-5. doi: 10.1016/j.tox.2013.07.014.
[2] DNA adducts caused by tamoxifen and toremifene in human microsomal system and lymphocytes in vitro. Hemminki,K., Widlak,P. and Hou,S.-M. (1995). Carcinogenesis, 16, 1661–1664.
[3] Major difference in the hepatocarcinogenicity and DNA adduct forming ability between toremifene and tamoxifen in female Crl:CD(BR) rats. GC Hard, MJ Iatropoulos, K Jordan, L Radi, OP Kaltenberg, AR Imondi, and GM Williams. Cancer Res., Oct 1993; 53(19): 4534-41.
[4] Apoptosis in toremifene-induced growth inhibition of human breast cancer cells in vivo and in vitro. Wärri AM, Huovinen RL, Laine AM, Martikainen PM, Härkönen PL. J Natl Cancer Inst. 1993 Sep 1;85(17):1412-8.
[5] Hormonal effects of toremifene in breast cancer patients. Számel I, Vincze B, Hindy I, Kerpel-Fronius S, Eckhardt S, Mäenpää J, Grönroos M, Kangas L, Sundquist H, Hajba A. J Steroid Biochem. 1990 Jun 22;36(3):243-7.
[6] Influence of toremifene on the endocrine regulation in breast cancer patients. Számel I, Hindy I, Vincze B, Eckhardt S, Kangas L, Hajba A. Eur J Cancer. 1994;30A(2):154-8.
[7] The effect of selective estrogen receptor modulator administration on the hypothalamic-pituitary-testicular axis in men with idiopathic oligozoospermia. Tsourdi E, Kourtis A, Farmakiotis D, Katsikis I, Salmas M, Panidis D. Fertil Steril. 2009 Apr;91(4 Suppl):1427-30. doi: 10.1016/j.fertnstert.2008.06.002. Epub 2008 Aug 9.




